I am trying to figure out how the reactivity series comes about. My understanding is that elements with a higher electronegativity will be more reactive than elements with a lower electronegativity, and that elements with a low ionization energy will be more reactive than elements with a high ionization energy.
Here is a chart of electronegativity (from Wikipedia):
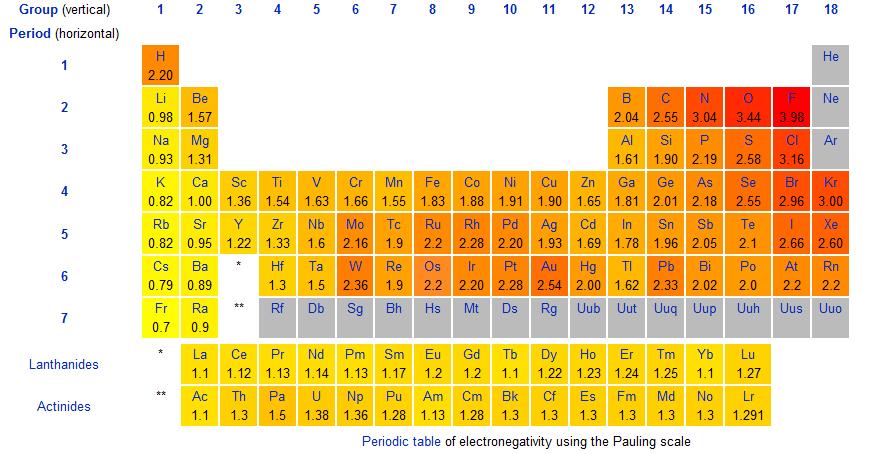
This shows electronegativity decreasing (as reactivity increases) down Group 1 and it also decreasing (as reactivity decreases) down Group 7.
Here is a chart of ionization energy:

This shows ionization energy decreasing (as reactivity increases) down Group 1 and it also decreasing (as reactivity decreases) down Group 7.
Neither electronegativity nor ionization energy can predict both reactivity trends, so what is going on?
No comments:
Post a Comment