This reversible reaction occurs in room condition:
$$\ce{2NO2 <=> N2O4}\quad(\Delta H=-57.23\mbox{ kJ/mol)}$$
This is the Lewis structure of $\ce{NO2}$ (courtesy Ben Mills via Wikipedia):
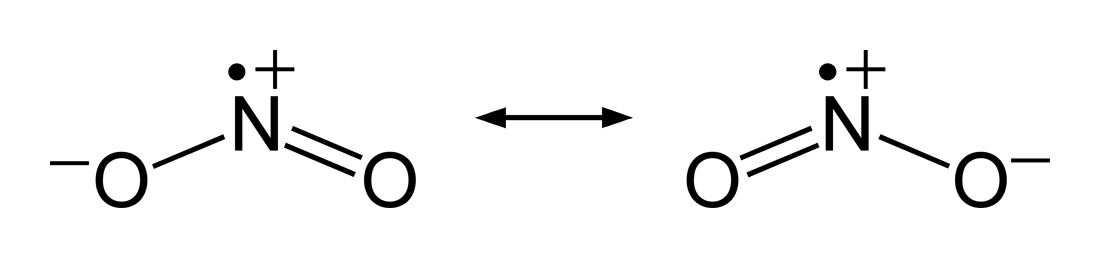
This is the Lewis structure of $\ce{N2O4}$ (courtesy ChemSpider):

To me, it seems that no bonds need to be broken for the forward reaction ($\ce{2NO2 -> N2O4}$) to occur, which is strange indeed.
Answer
No bonds are broken, but there are several isomers of N2O4. The unpaired electron can be on either the N or the O, for each NO2 molecule. The long bond between the monomers involves these two unpaired electrons.
See sections 7-1 to 7-3 of Bonding in Electron-Rich Molecules: Qualitative Valence-Bond Approach via Increased-valence Structures for more information.
According to First-Principles Study of the Role of Interconversion Between NO2, N2O4, cis-ONO-NO2, and trans-ONO-NO2 in Chemical Processes J. Am. Chem. Soc., 2012, 134 (31), pp 12970–12978:
there is no barrier to the bonding of two monomers to form the planar isomer with the N-N bond.
No comments:
Post a Comment